
Share Important Moment of MileCell Bio with You
2026.03.14
The success of clinical-grade cell therapies depends on safe and reliable cell freezing. While DMSO in traditional media protects cells, it can cause patient side effects and complicate manufacturing.
Kryogene® Cell Freezing Media – DMSO Free is the innovative solution. It contains no DMSO, removing toxicity concerns and regulatory hurdles, smoothing the path from lab to clinic.
Our media is not only safe but also high-performing. Data (Fig 1A) shows that Adipose-derived MSCs (AD-MSCs) frozen with our product maintain excellent viability, matching or exceeding DMSO-based competitors.
More importantly, cell quality and function are fully preserved. Surface marker expression remains normal (Fig 1B), and the cells retain their strong ability to differentiate into fat, bone, and cartilage tissues after thawing (Fig 1C), proving their therapeutic potential is intact.
Manufactured under cGMP standards with a defined, animal component-free formula, our product minimizes batch variation and immunogenic risk. The single-use manufacturing process prevents cross-contamination, and endotoxin levels are tightly controlled (≤ 0.5 EU/mL), meeting strict clinical-grade requirements.
Choosing Kryogene® DMSO-Free Freezing Media means choosing a safer, simpler, and more compliant path for your cell-based product development. It protects your cells and safeguards your clinical progress.
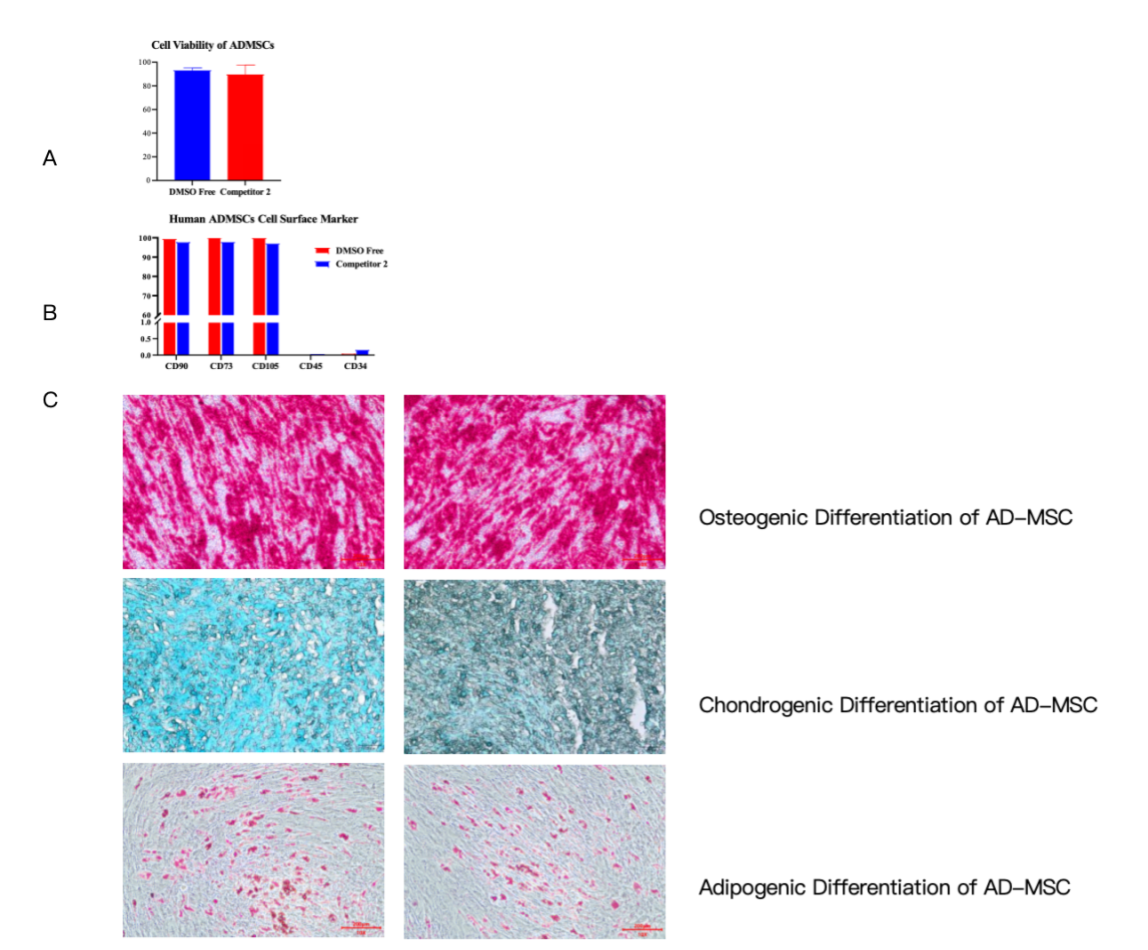
Fig 1. In Vitro Functional Assessment of Adipose Tissue-Derived MSCs cryopreserved with Kryogene Cell Freezing Media-DMSO Free and Competitive product.
(A) Post-thaw cell viability of AD-MSCs.
(B) Cell surface marker expression of AD-MSCs post-thaw.
(C) Post-thaw Adipogenic/Osteogenic/Chondrogenic differentiation of AD-MSCs.
