
Share Important Moment of MileCell Bio with You
2026.02.20
ü High post-thaw viability
ü Morphological integrity and attachment efficiency
ü Individual and pooled donor lots available
ü Functional Validition:Transporter Qualified, 3D Spheroids Qualified, ASGPR Qualified
ü Comprehensive Species Coverage:Mouse, Rat, Monkey, Dog, Cat, Rabbit, Minipig, Hamster, etc.
ü Well-Stocked, Fresh & Viable Inventory
ü Customizable: Gender, Species, Pack Size, Batch Size, Cell Specification
Primary hepatocytes in sandwich culture is created by coating the cell monolayer with collagen I or Matrigel on both sides, inducing the re-establishment of cellular polarity and the formation of bile canaliculi-like networks, which provides an in vivo-like model for studying biliary efflux and uptake drug transporters, evaluating prolonged hepatic metabolism, drug-drug interactions (DDI), and mechanisms of chronic drug-induced liver injury (DILI).
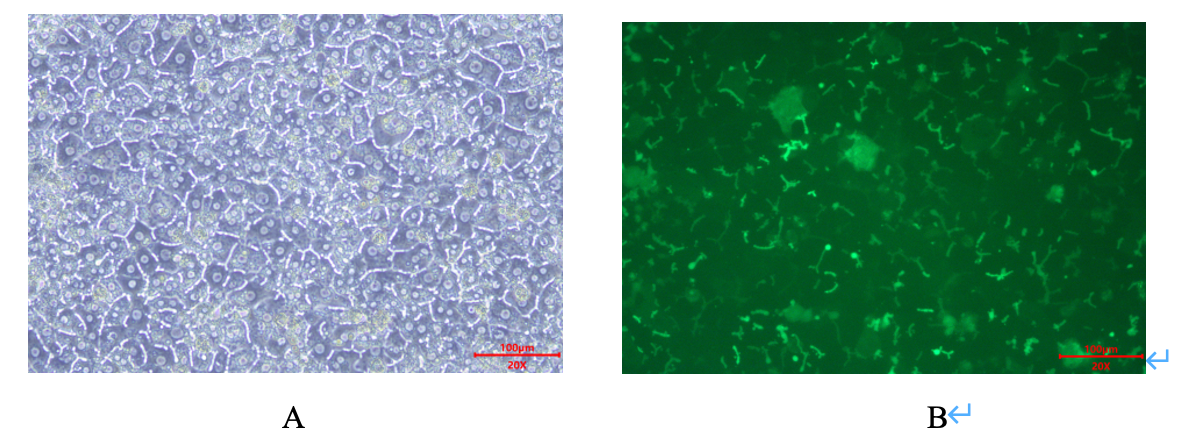
Figure 1:Sandwich culture of SD Rat Plateable Hepatocytes demonstrates 100% confluency and forms bile canalicili on day 3 (A). The staining of CDFDA uptaken into Bile Canaliculi (B).
3D Spheroids Qualification
Three-dimensional culture maintain hepatocyte viability while preserving physiologically relevant phenotypes, gene expression, and protein profiles. Compared to traditional monolayer culture systems, spheroid models require fewer cells, offer greater technical simplicity, and can be readily adapted for high-throughput applications.

Figure 2:3D Spheroid formation of Cynomolgus Monkey Hepatocytes with 3,000 cells. After 8 days of culture, cells form spheroids with a diameter of approximately 250-300μm.
MileCell provides tailored product solutions with flexible choices in Species, Gender, Donor Pooled, Cell Specifications.
At MileCell, we are committed to providing both Hepatocytes and Non-parenchymal cells (NPCs) to meet the specific needs of a wide range of scientific research applications, including drug development and hepatotoxicity testing, inflammation, fibrosis, and tissue regeneration. Under optimized tissue harvesting protocols, enhanced cell isolation techniques significantly improve cells viability, purity, and structural integrity, which—together with diverse post-thaw functional characterization—collectively set a new industry benchmark for cells quality and performance.