
Share Important Moment of MileCell Bio with You
2026.02.20
Liver subcellular fractions, rich in metabolic enzymes derived from the endoplasmic reticulum, are key in vitro systems for evaluating drug metabolism. These fractions are widely used to study cytochrome P450 inhibition, metabolic stability, phenotyping, and metabolite characterization. This versatile model provides critical insights into the metabolic behavior of drug candidates.
▪ Quality Assurance
All batches undergo comprehensive quality control procedures to ensure product integrity and batch-to-batch consistency.
▪ Comprehensive Characterization
Characterization of key CYP450 and Phase II enzyme activities, ensuring exceptional metabolic performance and specificity for consistent and reliable results.
▪ Scale Production
Large lots derived from donor pools of liver subcellular fractions to minimize lot-to-lot variation and ensure long-term availability across experimental replicates.
▪ Comprehensive Species Coverage
Mouse, Rat, Monkey, Dog, Cat, Rabbit, Minipig, Hamster, Guinea pig, etc.
▪ Well-Stocked, Fresh & Viable Inventory
▪ Customizable: Gender, Species, Vial Size, Batch Size, etc.
Metabolic Stability
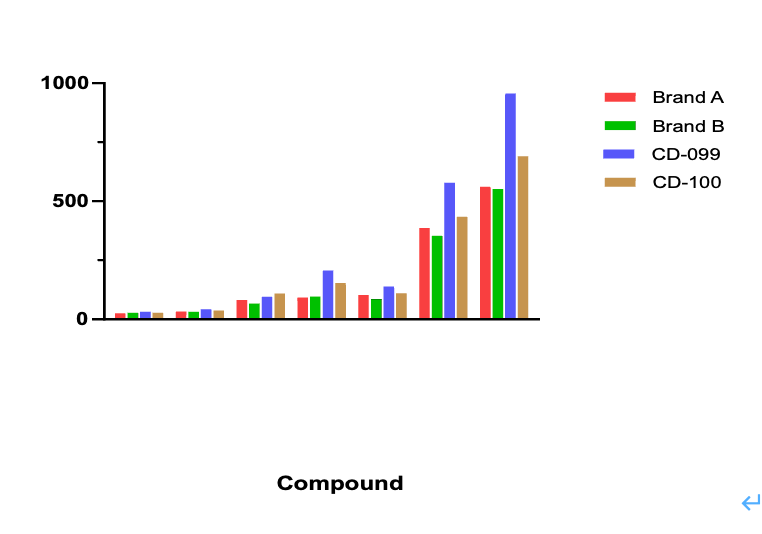
Figure 1: Representative stability experiments conducted on three branded CD-1 mouse liver microsomes. In comparison to competitive products from Brand A and Brand B, the MileCell liver microsomes (CD-099、CD-100) exhibit higher intrinsic clearance (Clint, Bar chart) for the seven compounds tested.
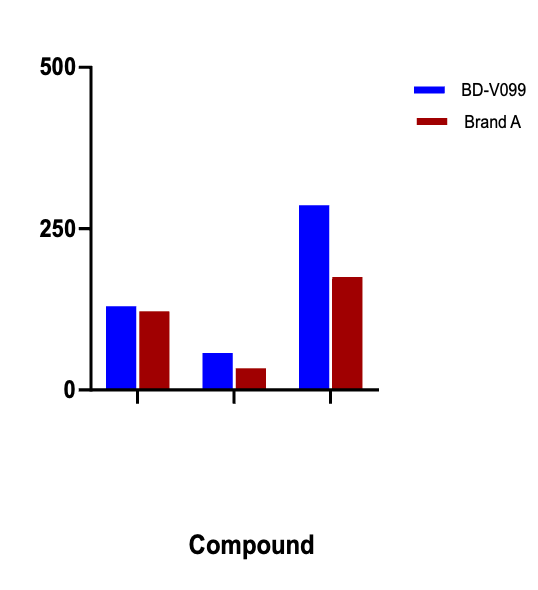
Figure 2: MileCell Beagle Dog Liver S9 (Lot # V099) shows higher clearance rates for Testosterone, Verapamil, and Raloxifene compared to competitive brand.