
Share Important Moment of MileCell Bio with You
2025.04.03
At Kryogene (a subsidiary of MilesBio), we’re advancing cell therapy by solving one of its biggest challenges: reliable cryopreservation. Our Kryogene™ Cell Freezing Media-CGT (FDA DMF 040563) is engineered to safeguard cell integrity for critical applications, including human umbilical cord-derived mesenchymal stem cells (MSCs).
Why Kryogene™ Stands Out:
üRegulatory-Compliant: Manufactured under cGMP standards with USP/ChP - grade materials.
üAnimal Component-Free: No serum, proteins, or animal-derived ingredients - ideal for clinical use.
üProven Performance: Optimized for high viability, functional retention, and scalability.
Data Spotlight: MSC Cryopreservation Excellence
In rigorous testing, MSCs frozen with Kryogene™ demonstrated:
1. High Post-Thaw Viability: >90% cell survival (comparable to fresh cells), (see Fig. 1).
2. Uncompromised Differentiation Capacity: Osteogenic, chondrogenic, and adipogenic differentiation potential fully retained (see Fig. 2).
3. Stable Phenotype: Surface markers (CD73, CD90, CD105) maintained post-thaw, ensuring therapeutic consistency(see Fig. 3).
Competitive Edge:
Kryogene™ outperforms conventional DMSO-based media by eliminating batch variability and cytotoxicity risks. Whether you’re scaling CAR-T therapies or banking MSCs, our formulation delivers reproducible, clinic-ready results.
Join the Innovation
[https://www.milecell-bio.com/] or contact our team at info@milecell-bio.com for samples and data packages.
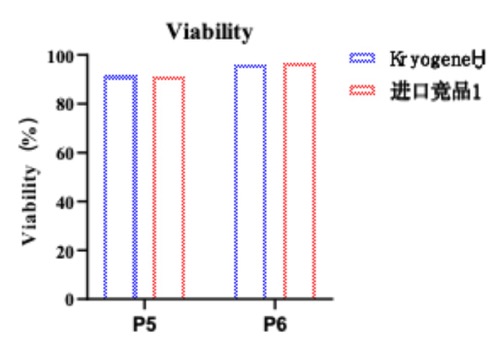
Fig. 1 Graphical comparison of post-thaw viability (%) vs. competitor products.
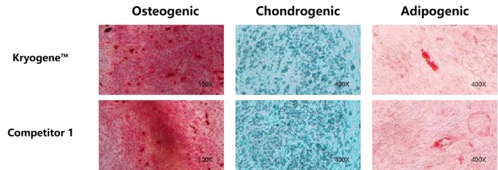
Fig. 2 Side-by-side differentiation images of MSCs (osteogenic/chondrogenic/adipogenic) post-thaw.
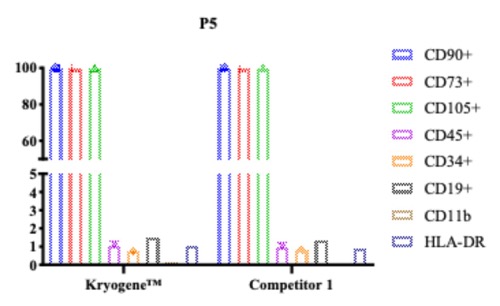
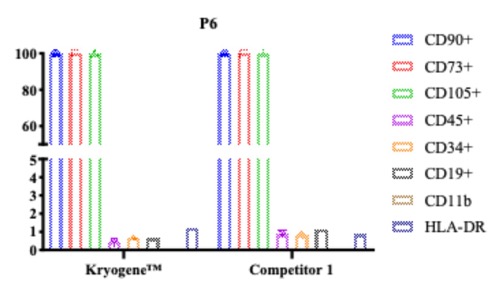
Fig. 3 Flow cytometry plots comparing MSC surface marker expression post-freezing.
